
http://www.physics.usyd.edu.au/hienergy/index.php/The_Fundamental_Particles | Download Scientific Diagram

SOLVED:Consider an atom of 10 $\mathrm{B}$ . (a) How many protons, neutrons, and electrons does this atom contain? (b) What is the symbol of the atom obtained by adding one proton to $^{

True or false? Adding one neutron to the nucleus of an atom converts it to an isotope of the same element. | Study.com
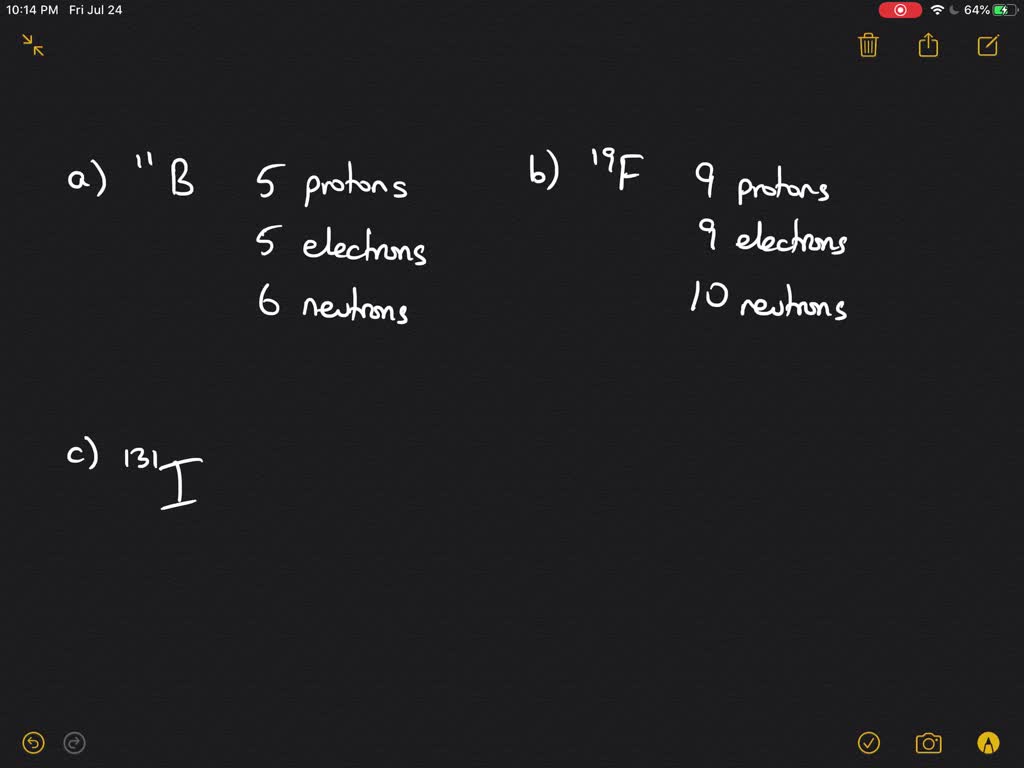
SOLVED:3 Consider an atom of 10B. (a) How many protons, neutrons, and electrons does this atom contain? (b) What is the symbol of the atom obtained by adding one proton to 10B? (

Adding neutrons to synthetic atoms drastically alters shape of their nuclei, affects their stability

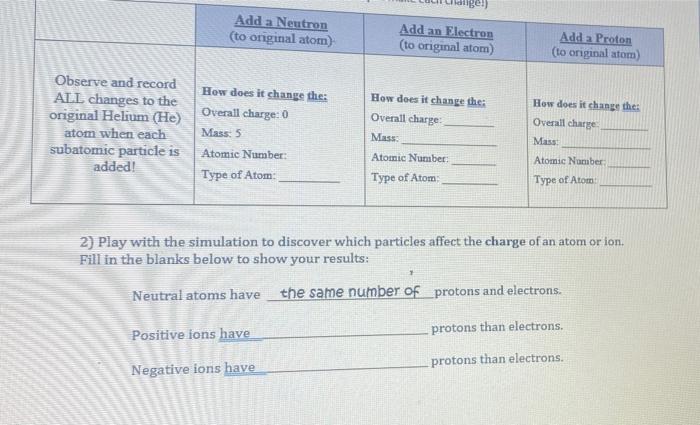
Nuclear energy Summary of period 1. Parts of the atom Mass (A.M.U)Charge Proton1+1 Neutron10 Electron1/1830 Add/subtract a proton: creates a different. - ppt download
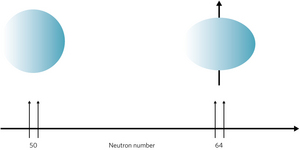
Adding neutrons to synthetic atoms drastically alters shape of their nuclei, affects their stability

Protons Neutrons Electrons Isotopes - Average Mass Number & Atomic Structure - Atoms vs Ions - YouTube







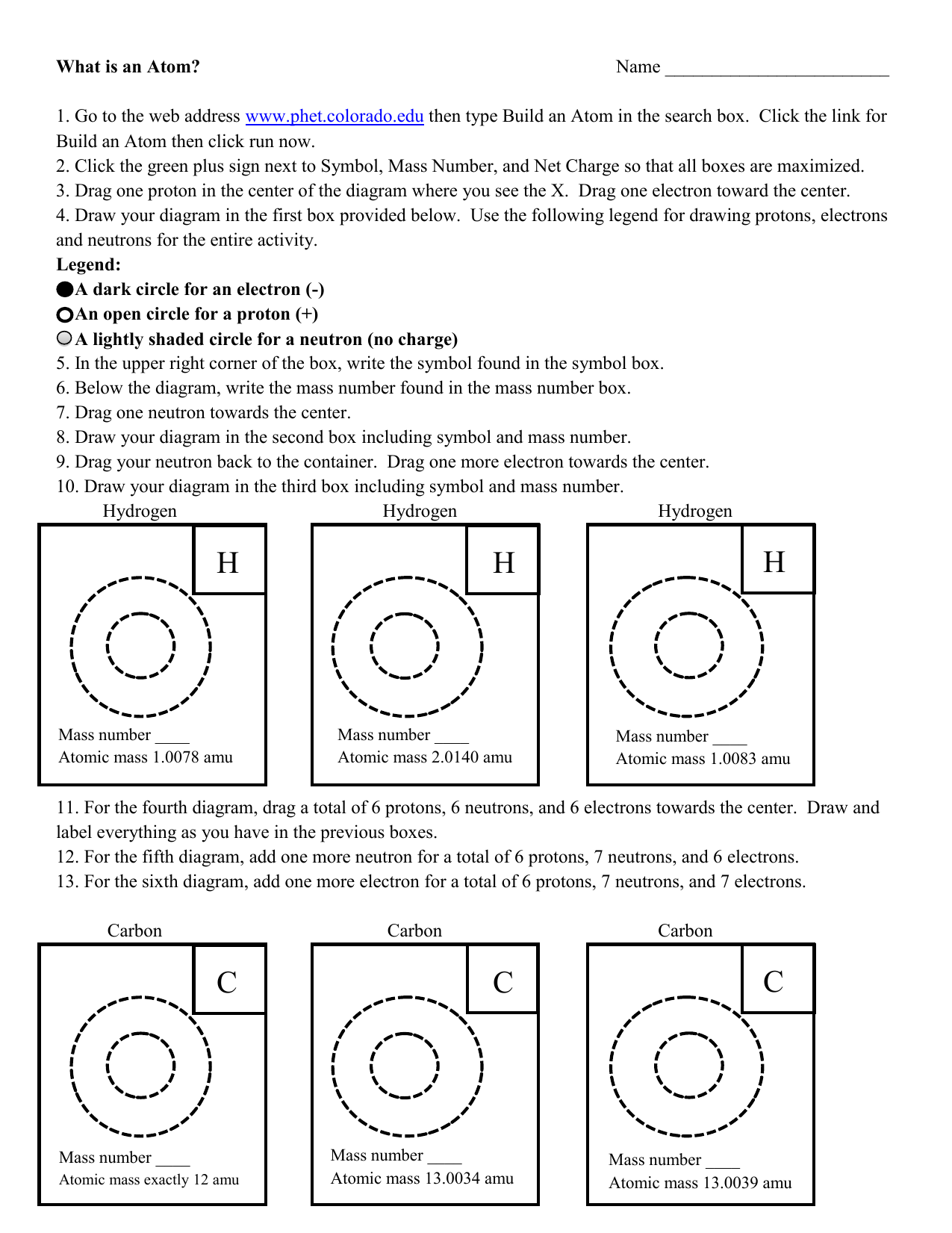

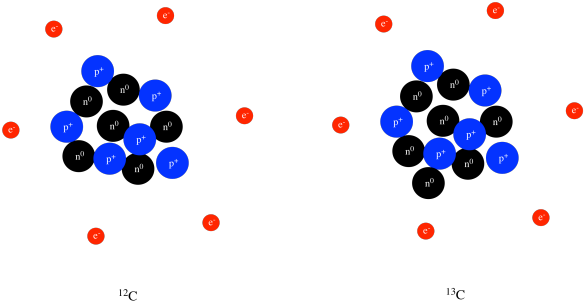
/GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)

