Balance the following equation by oxidation number method. PbCrO4 + H2SO4 + FeSO4→ Fe2(SO4)3 + PbSO4 + Cr2(SO4)3 + H2O

PDF) Aqueous Solutions of Cr(III) Sulfate: Modeling of Equilibrium Composition and Physicochemical Properties
![The equivalent weight of Cr2(SO4)3 [mot wt. = M] in the following reaction is Cr2(SO4)3 + H2O2 + NaOH → Na2CrO4 + Na2SO4 + H2O The equivalent weight of Cr2(SO4)3 [mot wt. = M] in the following reaction is Cr2(SO4)3 + H2O2 + NaOH → Na2CrO4 + Na2SO4 + H2O](https://d1hj4to4g9ba46.cloudfront.net/questions/1305019_698125_ans_e9f07e9b3a3e45088aea1d3aa5aa36ea.jpg)
The equivalent weight of Cr2(SO4)3 [mot wt. = M] in the following reaction is Cr2(SO4)3 + H2O2 + NaOH → Na2CrO4 + Na2SO4 + H2O
Balance the following equations by oxidation number method 1. K2Cr2O7 + KI + H2SO2 → K2SO4 + Cr2(SO4)3 + I2 + H2O - Sarthaks eConnect | Largest Online Education Community

PDF) Determination of the Diffusion Coefficient of the Green and Violet Isomers of Cr2(SO4)3 in Aqueous Solution

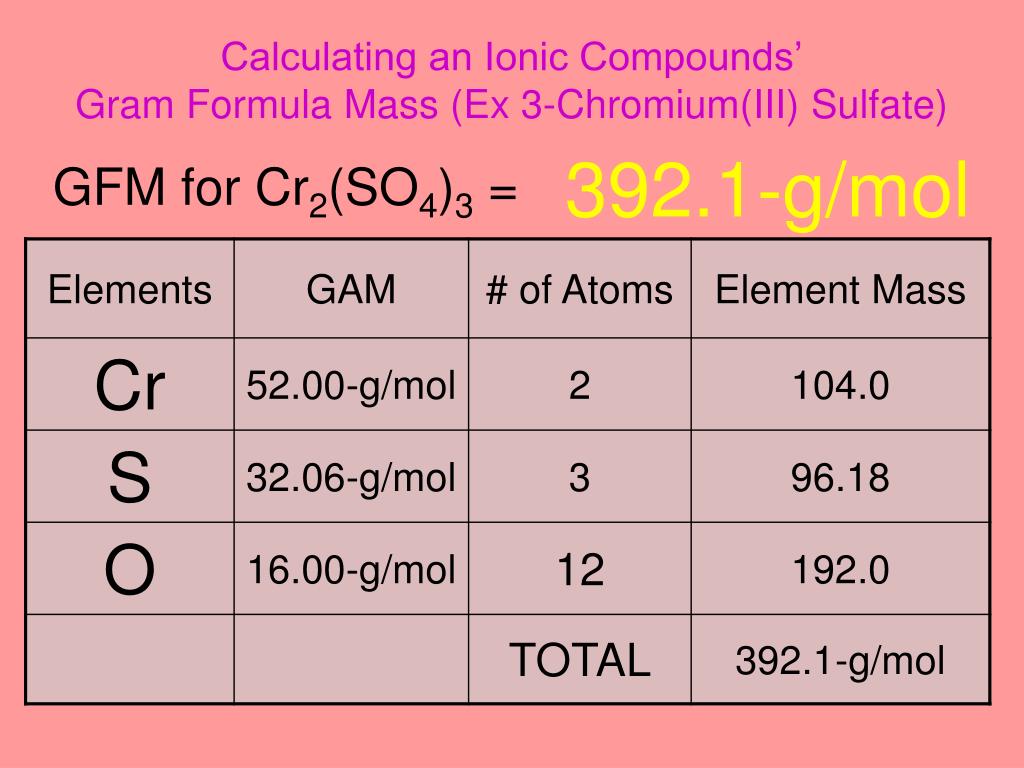
SOLVED:Complete the following for the compound chromium(III) sulfate formula Crz(SO43 atom number in formula chromium sulfur oxygen