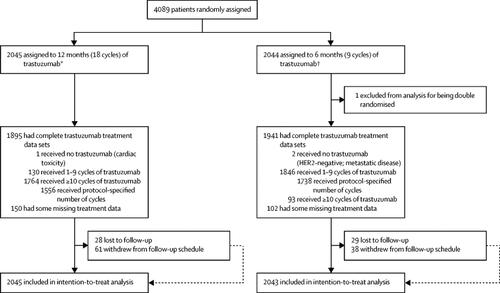
Ioannis MAMOUNAKIS | Research Associate | Doctor of Philosophy Computer Engineer and Informatics | The Centre for Research and Technology, Hellas, Thessaloníki | Chemical Process and Energy Resources Institute (CPERI)
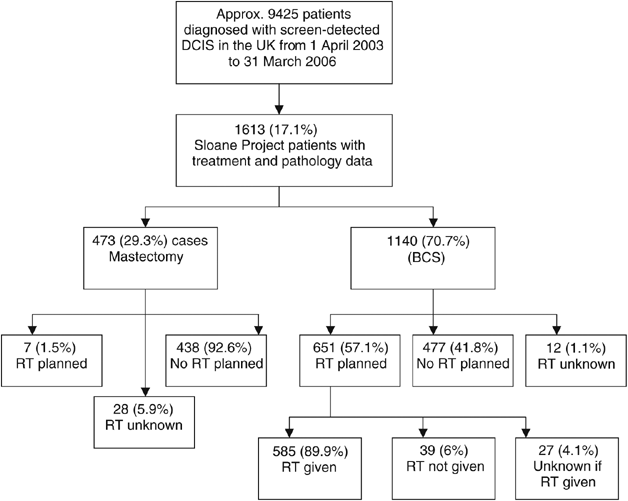
Radiotherapy following breast-conserving surgery for screen-detected ductal carcinoma in situ : indications and utilisation in the UK. Interim findings from the Sloane Project | British Journal of Cancer
ΜΑΜΟΥΝΑΣ ΙΩΑΝΝΗΣ | Διδάκτωρ Της Ψυχιατρικής Του Πανεπιστημίου Αθηνών - Άγχος - Αγχώδεις Διαταραχές – Κατάθλιψη - Αϋπνία & Άλλες Διαταραχές Ύπνου - Γνωσιακή Ψυχοθεραπεία - Θεραπεία Οικογένειας – Ζεύγους-Ψυχαναγκασμοί - Βουλιμία - Ανορεξία, Ψυχίατροι ...